52 yrs female with HTN had agonising abdominal pain for a day...Admitted to a surgeon's place....X-ray , USG: Did not show any abnormality and TLC was near normal..
but severe pain in abdomen----surgeon Doctor promptly advised for CECT abdomen suspecting MAT(Mesenteric Arterial thrombosis)..
And was right in Diagnosis
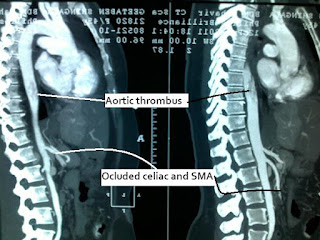
pt had a very bad CECT Picture:
Immediately underwent Mesenteric angio and Inj Heparin was started in infusion...
Fortunately by angiography Hepatic artery, spleenic artery, IMA and celiac was opened...pulse injection of PAPAVERIN Was given in vessels...and catheter was left in SMA for cont infusion...
Next day pt had relief in Pain but started developing metabolic acidosis in ABG (FIRST MARKER OF BOWEL ISCHEMIA --Even before pulse and BP Alteration )
Repeat ABG in afternoon showed marked drop in Ph and severe metabolic acidosis...
Heparin stopped, Protamine given and taken up for surgery...
fortunately she is saved of
LIVER, SPLEEN,KIDNEY PROXYMAL 2 FEET OF SMALL BOWEL AND ASCENDING COLON ONWARDS LARGE BOWEL....
ALL BECAUSE OF EARLY SUSPECTING , CECT ( by primary consultant within 12hrs ) & INTERVENTION (by Intervention radiologist) AND TIMELY ACTION (surgery) WHICH IS REALLY DIFFICULT AND VERY LESS OFTEN POSSIBLE...but a team effort made it possible...
Pt was saved going into septecemia and shock and now on re-feeding of stoma contents with oral diet and antico-agulants....ambulant in ward ..(POD -7)
now will require high nutritional supplimentation....and close observations...fingers are still crossed !!! lets hope for full recovery.....
www.gisurgerysurat.com/
www.drkeyurbhatt.in/
www.sidshospital.com
Dr. Keyur Bhatt - Best
Gastro Surgeon
Dr. Keyur Bhatt- Best GI
Surgeon
Dr. Keyur Bhatt - Best
Gastro Surgeon
Dr. Keyur Bhatt- Best GI
Surgeon - Dr Keyur Bhatt - Best Gastro Surgeon
Dr. Keyur Bhatt- Best GI
Surgeon - Dr Keyur Bhatt - Best Gastro Surgeon
Dr Keyur
Bhatt- Best GI Surgeon